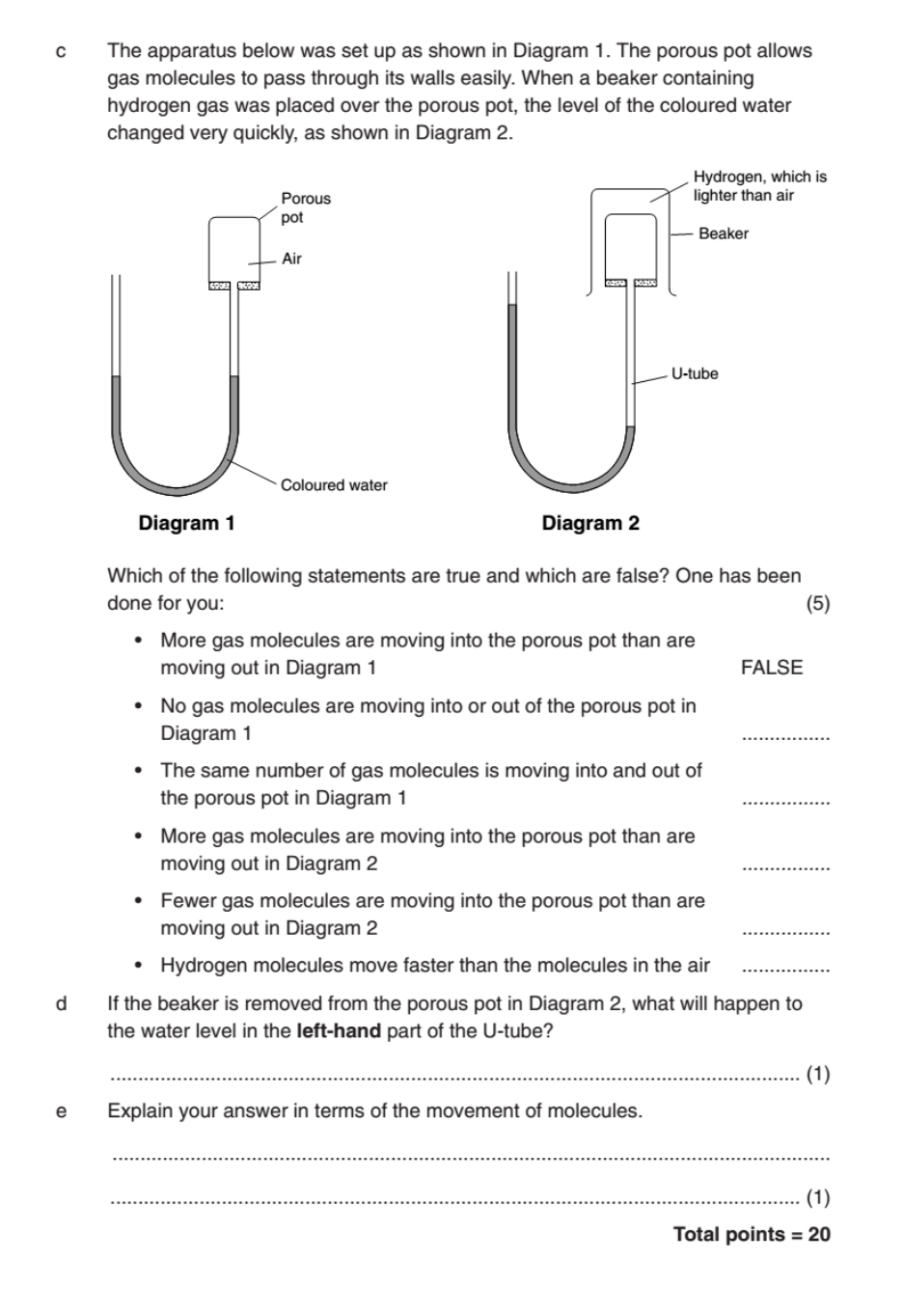
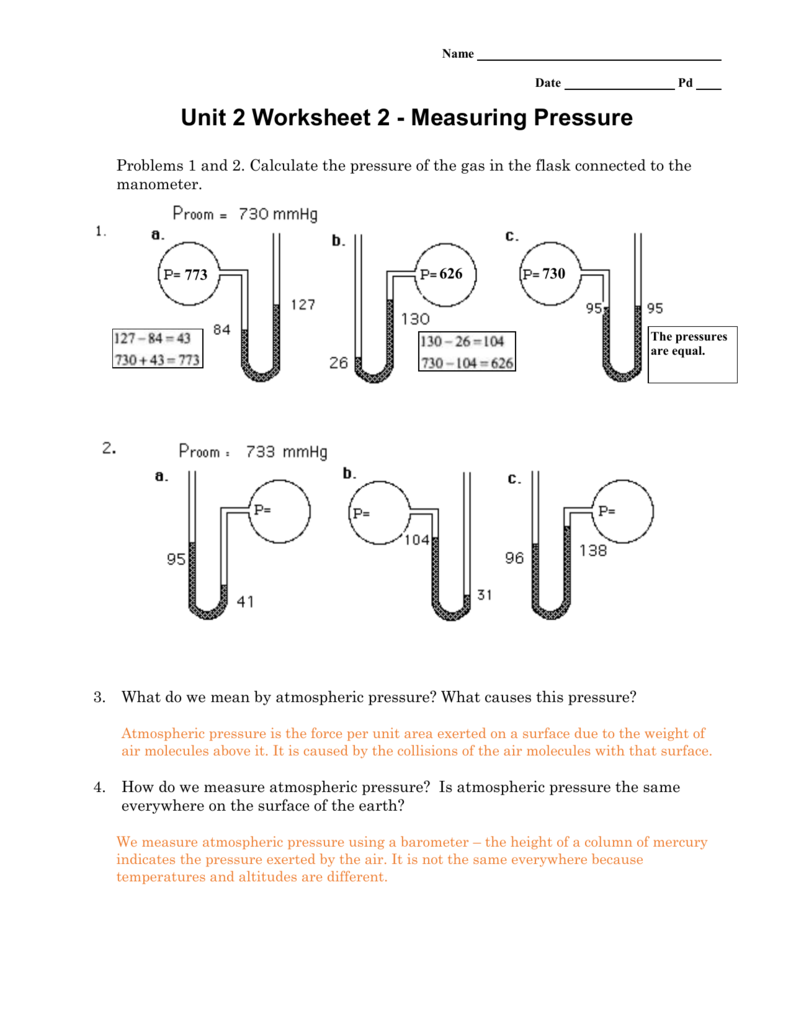
Problems and 2. Calculate the pressure of the gas in the flask connected to the manometer. Unit Worksheet – Measuring Pressure. A device used to measure the pressure of a gas is called a manometer. What do we mean by.

A sample of gas is. A manometer is a device that measures the pressure of a gas in an enclosed container. Chem Worksheet 13- 2. Gas pressure is equal. Jul More in gas laws unit.
Clinical Guidelines Nursing Pressure Injury Prevention And. A gas with a volume of 4. L at a pressure of. The gas is heated 77°C at. If I raise the temperature to 350. K and lower the pressure to. L the pressure of each gas is equal to the number of moles. Subscript ( ) = new condition or final condition. Name: Match the variables used to describe gases to the correct unit. C, what is the pressure inside the container? It is not safe to put aerosol.
State the values for standard temperature: standard pressure : 3. Convert the following pressures: a. A hydrogen gas volume thermometer has a volume of 100. If a gas originally occupying 6. If this can is lal thrown unto a fire.
C, how many moles of gas do I have? If I have an unknown quantity of gas at a pressure of 1. In a thermonuclear device, the pressure of 0. An introduction to the relationship between. Perform the following calculation, and match the question to the individual gas law.
Use the pressure unit of mm Hg. A flashbulb of volume 2. Ogas at a pressure of 2. How many moles of oxygen gas does. Lat standard pressure to 740. Experiment Pressure (atm) Volume (L).
When the stopcock between the two flasks is opened and the gases are.